|
They can predict properties such as stability, boiling point, and conductivity. Using principles of physics, chemists can predict how atoms will react based upon the electron configuration. The electron configuration is the orbital description of the locations of the electrons in an unexcited atom. These atomic weights should be considered provisional since a new isotope with a longer half-life could be produced in the future. 
The IUPAC convention is to list the atomic weight of the longest-lived isotope in the periodic table. However, for man-made trans-uranium elements there is no "natural" abundance. For naturally-occurring elements, the atomic weight is calculated from averaging the weights of the natural abundances of the isotopes of that element. The average number of neutrons for an element can be found by subtracting the number of protons (atomic number) from the atomic mass.Ītomic weight for elements 93-118. Though individual atoms always have an integer number of atomic mass units, the atomic mass on the periodic table is stated as a decimal number because it is an average of the various isotopes of an element. The standard atomic weight is the average mass of an element in atomic mass units ("amu"). Click here for a list of the elements and their symbols. Typically, a symbol is the truncated name of the element or the truncated Latin name of the element. The atomic symbol is one or two letters chosen to represent an element ("H" for "hydrogen," etc.).  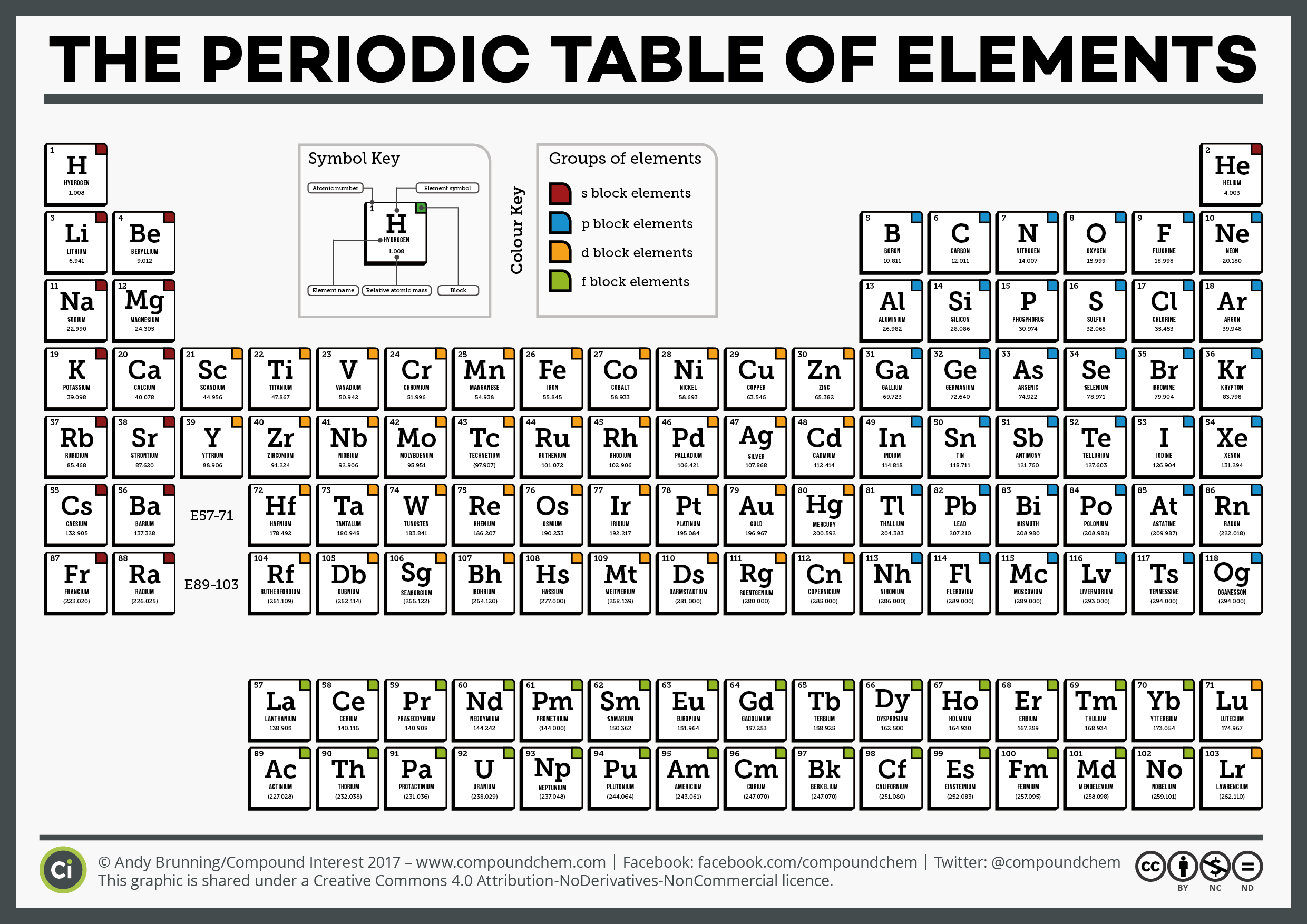
The number of protons in an atom also determines the chemical behavior of the element. The number of protons in an atom is referred to as the atomic number of that element. For example carbon atoms have six protons, hydrogen atoms have one, and oxygen atoms have eight. The number of protons in an atom defines what element it is. When you open any file of an element in the periodic table, you will find a small table with some basic information about that element.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
